I. 製品概要
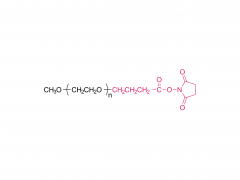
N-フルオレニルメトキシカルボニル-ポリエチレングリコール-プロピオン酸は、構造的に精密で明確な切断可能なPEGリンカー試薬の一種です。Fmoc保護基と末端カルボン酸が様々な長さのポリエチレングリコール鎖を介して連結されたこのシリーズは、生体共役、創薬、およびケミカルバイオロジー研究のための多用途な分子ツールを提供します。
II. 構造的特徴
Fmoc保護アミン:
穏やかな塩基性条件下(通常は20%ピペリジン/DMF)で除去でき、反応性のアミノ基を露出させて後続の結合反応に利用できる。
末端カルボン酸:
(例えばEDCやDCCを用いて)活性化することで、アミノ基含有化合物と安定なアミド結合を形成することができる。
調整可能なPEG鎖長:
親水性、立体障害、スペーサー間距離に影響を与える。
優れた物理化学的特性:
水溶性と生体適合性の向上
免疫原性の低下と非特異的相互作用の最小化
薬物分子の安定性と半減期の改善
III. 主な応用例
1. PROTACテクノロジー
PROTAC(タンパク質分解標的キメラ)分子の構築において重要な役割を果たす。
リンカー機能:
空間最適化:
E3リガーゼリガンドと標的タンパク質リガンド間の最適な空間配置のために、適切な長さと柔軟性を提供します。
物理化学的変調:
水溶性を高め、細胞透過性を改善し、親水性/疎水性のバランスを整えることで、膜透過性と生体利用率に影響を与える。
安定性の向上:
酵素分解を抑制し、生体環境におけるPROTACの安定性を向上させます。
構造的多様性:
異なるPEG鎖長を用いて分解効率を最適化するためのスクリーニングプラットフォームを提供する。
PROTAC合成における具体的な用途:
E3リガーゼリガンドの修飾:
Fmoc-PEG-COOHを介してE3リガーゼリガンド(例えば、サリドマイド、VHL、またはセレブロンリガンド)をPROTACコアに連結します。
標的タンパク質リガンドの機能化:
標的タンパク質リガンドをPROTACリンカーユニットに結合させる。
二官能性リンカー:
PROTAC分子の両端に反応性基を導入することで、非対称な連結構造を構築する。
2. ペプチドおよびタンパク質化学
固相ペプチド合成:
ペプチド合成中に特定の官能基を導入するための、切断可能なリンカーとして機能する。
タンパク質の標識化と修飾:
蛍光色素、ビオチン、またはその他の機能性分子を特定のタンパク質部位に結合させる。
抗体薬物複合体(ADC)の開発:
細胞毒性薬と抗体との間の切断可能なリンカーとして機能する。
3. 材料表面の機能化
バイオセンサーの表面修飾
ナノ粒子の機能化
ハイドロゲル材料の改質
4. 低分子医薬品の開発
プロドラッグ設計:薬物の溶解性と薬物動態特性を向上させる。
標的指向型薬物送達システムの構築。